Electrochemistry

- Galvanic Cell
Oxidation is defined as the addition of oxygen to a substancethe loss of hydrogen from a substancethe...
- Electrochemistry : 10.2 Nernst Equation
NERNST EQUATION Cell potential (E°cell) under any condition Ecell = E°cell – (RT/nF) ln Q R: universal gas constant. Q...
- Thermochemistry : 9.3 Born-haber Cycle
First Ionization Energy (IE1)Energy required for 1 mol of gaseous atom to lose 1 mol of electrons. Affinity Electron (EA) Energy change that occurs when 1 mol of gaseous atom gains 1 mol of electrons. Lattice EnergyEnergy change when 1 mol of solid...
- Thermochemistry : 9.2 Hess Law
Hess's Law of Heat Summation When reactants are converted to products, the change in enthalpy is the same whether the reaction take step in one step or in a series of step. ΔH1 =ΔH2 + ΔH3 Application Using Hess's...
- Group Member Of This Blog
Group 1 ~Thermochemistry~ Group Leader : Khor Sheng Yau Muhammad Amirul Syafiq Bin Azudin ...
Electrochemistry
Thermochemistry : 9.1 Concept of Enthalpy
Heat
-The energy transferred between a system and surrounding.
System
-Part of universe whose change we are going to observe.
Surrounding
-The rest of the universe outside the system.
Exothermic
-Gives off heat.
-From the system to the surroundings.
-Ex : 2H2(g) + O2(g) → 2H2O(l) + energy
Endothermic
-Absorbs heat.
-From the surrounding to the system.
-Ex : energy + 2HgO(s) → 2Hg(l) + O2(g)
Enthalpy
-Used to quantify the heat flow into or out of system in a process that occurs at constant pressure.
Enthalpy Change, ΔH
-Heat given off or absorbed during a reaction at constant pressure.
-ΔH = H(Products) – H(Reactants)
-Exothermic : H(Products) < H(Reactants)
-Endothermic : H(Products) > H(Reactants)
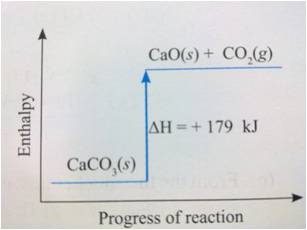
Enthalpy/Energy Profile Diagram

-Energy profile diagram for an exothermic reaction.

-Energy profile diagram for an endothermic reaction.
Units of Energy
-S.I. unit : Joule(J) [1J = 1kgm2/s2]
-Older unit : Calorie(cal) [1cal = 4.184J]
Some Important Types of Enthalpy Change(ΔH)
-Ex : Heat of combustion(ΔH comb)
Heat of formation(ΔH f)
Heat of neutralization(ΔH neut)
-Unit : kJ/mol
Enthalpy of Combustion(ΔH comb)
-Heat released when 1 mole of a substance completely combusted in oxygen gas.
-Ex : C4H10(l) + 13/2O2(g) → 4CO2(g) + 5H2O(l) ΔH = ΔH comb
Enthalpy of Formation(ΔH f)
-Heat change when 1 mole of a compound is produced from its elements.
-Ex : K(s) + 1/2Br2(l) → KBr(s) ΔH = ΔHf
Enthalpy of Atomization(ΔHatom)
-Heat absorbed when 1 mole of gaseous atom is formed from its element.
-Ex : 1/2H2(g) → H(g) ΔH = ΔHatom
Na(s) → Na(g) ΔH = ΔHatom
Enthalpy of Neutralization(ΔH neut)
-Heat released when 1 mole of water formed when acid reacts with base.
-Ex : HCl(aq) + NaOH(aq) → NaCl(aq) + H2O(l) ΔH = ΔHneut
HNO3(aq) + 1/2Ba(OH)2(aq) → 1/2Ba(NO3)2(aq) + H2O(l) ΔH = ΔHneut
Enthalpy of Solution(ΔHsoln)
-Heat change when 1 mole of solute dissolves in solvent/water to form infinite dilute solution.
-Ex : NaCl(s) → Na+(aq) + Cl-(aq) ΔH = ΔHsoln
Enthalpy of Hydration(ΔHhyd)
-Heat released when 1 mole of gaseous ion is hydrated in aqueous solution.
-Ex : Na+(g) → Na+(aq) ΔH = ΔHhyd
Enthalpy of Sublimation(ΔHsublim)
-Heat absorbed when 1 mole of substance in solid state sublimed.
-Ex : I2(s) → I2(g) ΔH = ΔHsublim
CO2(s) → CO2(g) ΔH = ΔHsublim
K(s) → K(g) ΔH = ΔHsublime = ΔHatom
ΔH°rxn or ΔH°(kJ/mol)
-Standard enthalphy of reaction.
-the enthalpy change of a reaction carried out at standard state (1 atm)
-standard enthalpy of formation, ΔH°f
-standard enthalpy of combustion, ΔH°comb
Standard enthalpy of neutralization, ΔH°neut
Standard states
-Gas: 1 atm & behave ideally
-Aqueous solution: 1 M
-Pure substance (element/compound): Most stable form of substance at 1 atm and temperature (usually 25°C)
Standard states of pure substance: oxygen,O2, H2, Na(s), Br2, H2O(l), NaCl(s)
Specific Heat Capacity (c)
-Amount of heat (q) required to raise temperature of 1 gram of a substance by 1 K (1°C)
c=q/m
ΔT = Tfinal-Tinitial
-unit: Jg-1K-1/Jg-1°C-1
Molar Heat Capacity
-Amount of heat required to raise temperature of 1 mol of a substance by 1 K/1°C
-unit: JK-1/J°C-1
C=mc C=q/ΔT q=CΔT
Calorimeter
-Device used to measure the heat released (or absorbed) by a physical or a chemical process.
Ø Constant-pressure
l Coffee-cup calorimeter
Ø Constant-volume
l Bomb calorimeter
Bomb calorimeter
-To measure heat released in combustion reaction .
How bomb calorimeter works
-With continual stirring, the initial temperature of preweighed water bath is noted.
-The sample is ignited electrically.
-Heat released from the combustion reaction is transferred to the rest of the calorimeter.
-The maximum temperature is measured.
Calculation of ΔHrxn
qsys=qwater+qcal+qrxn
No heat enters/leaves the system
qsys=0
qrxn=-(qcal+qwater)
qwater=mcΔT
qcal=CcalΔT
Reaction at constant volume ΔH≈qrxn
Coffee-cup calorimeter
-To measure heat released in non-combustion reaction such as heat of neutralization &heat of solution.
-constant pressure calorimeter
-the process is opened to the laboratory atmosphere.
-no heat enters or leaves.
How coffee-cup calorimeter works
-The calorimeter consists of a known mass of water (or solution) in an insulated container equipped with a thermometer and a stirrer.
-Tinitial of water is measured
-The process takes place (addition of acid and base or soluble salt, etc)
-Contents are stirred.
-Tfinal of water is measured.
Calculation of ΔHrxn
qsys=qsoln+qcal+qrxn
No heat enters or leaves the system
qsys=0
qrxn=-(qsoln+qcal)
qsoln=mcΔT
qcal=CcalΔT
Reaction at constant P
ΔH=qrxn
Assume specific heat capacities &density same like water.
(4.184Jg-1°C-1) (1.00gmL-1)
Stoichiometry of thermochemical equation
CH4(g) + 2O2(g) → CO2(g) + 2H2O(l)
ΔH=-899.4kJ
-Sign=positive/negative
-Magnitude
Sign of ΔH
-Depends on reaction is exothermic or endothermic
-ve: exothermic +ve: endothermic
-A forward reaction has a opposite sign of the reverse reaction
Magnitude of ΔH
-Proportional to amount of substance reacting or produced.
2H2(g) + O2(g) → 2H2O(l) ΔH=-572kJ
H2(g) + 1/2O2(g) → H2O(l) ΔH=-286kJ
-286kJ if thermodynamically equivalent to 1 mole of H2
-286kJ is thermodynamically equivalent to 1/2 mole of O2
-286kJ is thermodynamically equivalent to 1 mole of H2O
- Galvanic Cell
Oxidation is defined as the addition of oxygen to a substancethe loss of hydrogen from a substancethe...
- Electrochemistry : 10.2 Nernst Equation
NERNST EQUATION Cell potential (E°cell) under any condition Ecell = E°cell – (RT/nF) ln Q R: universal gas constant. Q...
- Thermochemistry : 9.3 Born-haber Cycle
First Ionization Energy (IE1)Energy required for 1 mol of gaseous atom to lose 1 mol of electrons. Affinity Electron (EA) Energy change that occurs when 1 mol of gaseous atom gains 1 mol of electrons. Lattice EnergyEnergy change when 1 mol of solid...
- Thermochemistry : 9.2 Hess Law
Hess's Law of Heat Summation When reactants are converted to products, the change in enthalpy is the same whether the reaction take step in one step or in a series of step. ΔH1 =ΔH2 + ΔH3 Application Using Hess's...
- Group Member Of This Blog
Group 1 ~Thermochemistry~ Group Leader : Khor Sheng Yau Muhammad Amirul Syafiq Bin Azudin ...
