Electrochemistry
- Redox Equations
← Back to Electrochemistry Write balanced half-equations for the two reacting species. Multiply each half-equation to make the number of electrons equal. Combine the two half-equations. Simplify if necessary. Change...
- Oxidation Number
← Back to Electrochemistry Oxidation numbers are arbitrary numbers assigned to atoms to describe their relative state of oxidation or reduction i.e. how oxidised or reduced they are. They are assigned according to an arbitrary set of rules:...
- Practice Questions (a Levels) - Aqa
GCE AS/A Chemistry [Assessment and Qualifications Alliance (AQA)] You need Adobe Reader to read the files. Click here to download if you do not have it installed in your computer. Atomic Structure, Chemical Bonding & Periodicity Jun 2007Question Paper...
- Electrochemistry : 10.3 : Electrolysis Cell
VOLTAN CELL VS ELECTRIOLYSIS CELL . Voltaic cell :use a spontaneous reaction to generate electric energy. Electrolysis :use electric energy to drive non- spontaneous energy. VOLTAIC CELL. ELECTROLYTICelectrons generate...
- Electrochemistry : 10.1 Galvanic Cell
Electrochemistry Study of relationship between chemical change & electric work Oxidation Loss of electron by species accompanied byn an increase in oxidation number Ex: Reduction Gain electron by a species accompanied by a decrease number...
Electrochemistry
Which Half-Eqn to Use for H2O?
← Back to Electrochemistry
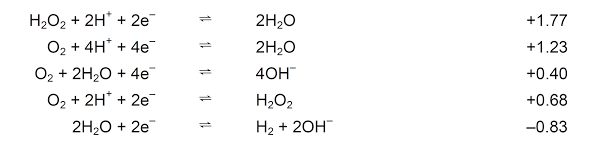
- In aqueous solutions (neutral)
H2O is oxidised to O2
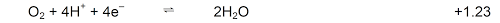
H2O is reduced to H2

- In acidic solutions (H+ present in significant amounts)
H2O is oxidised to O2
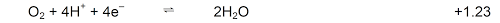
H+ is reduced to H2

- In alkaline solutions (OH– present in significant amounts)
OH– is oxidised to O2

H2O is reduced to H2

- In the presence of H2O2
H2O2 is oxidised to O2
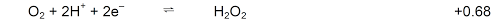
H2O2 is reduced to H2O
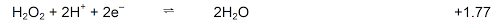
- Redox Equations
← Back to Electrochemistry Write balanced half-equations for the two reacting species. Multiply each half-equation to make the number of electrons equal. Combine the two half-equations. Simplify if necessary. Change...
- Oxidation Number
← Back to Electrochemistry Oxidation numbers are arbitrary numbers assigned to atoms to describe their relative state of oxidation or reduction i.e. how oxidised or reduced they are. They are assigned according to an arbitrary set of rules:...
- Practice Questions (a Levels) - Aqa
GCE AS/A Chemistry [Assessment and Qualifications Alliance (AQA)] You need Adobe Reader to read the files. Click here to download if you do not have it installed in your computer. Atomic Structure, Chemical Bonding & Periodicity Jun 2007Question Paper...
- Electrochemistry : 10.3 : Electrolysis Cell
VOLTAN CELL VS ELECTRIOLYSIS CELL . Voltaic cell :use a spontaneous reaction to generate electric energy. Electrolysis :use electric energy to drive non- spontaneous energy. VOLTAIC CELL. ELECTROLYTICelectrons generate...
- Electrochemistry : 10.1 Galvanic Cell
Electrochemistry Study of relationship between chemical change & electric work Oxidation Loss of electron by species accompanied byn an increase in oxidation number Ex: Reduction Gain electron by a species accompanied by a decrease number...
